|
All batteries have some tendency to freeze, but those with full charges are less susceptible. Batteries that become cold have less of their energy available, and some attempt to keep a battery warm before use is worthwhile. Several C cells might be found in large models. 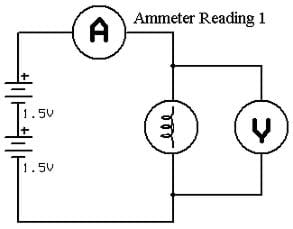
Most portable stereo-cassette players operate from two, three or four AA batteries (3 to 6 volts total). They may be found in mass-produced devices such as household smoke alarms. Silver oxide (1.5 V) and mercury (1.4 V) batteries are very good where nearly constant voltage is described at low currents for long periods. Numerous intercalated cathode materials have. N: (0.80 Ah) × (1.5 V) × (3600 s) = 2,700 J Updated On- 2023 Are There 1.5 V Rechargeable Batteries As an Amazon Associate, I Earn From Qualifying Purchases. Due to the abundance of sodium in nature, sodium-ion batteries (SIBs) have attracted widespread attention. The formula for determining cell energy is: The subject of whether or not 1.5 V batteries can be recharged in mobile devices is a common one. Furthermore, an alkaline electrolyte is used to provide a voltage for the battery, and silver-oxide and zinc are commonly used in the electrode. Advantages include high energy output, reliability, long shelf life, and superior low temperature performance. The 1.5 V batteries offer a great energy-to-weight ratio and have a voltage of 1.5V. Alkaline MnO 2 are very popular, multi use batteries. Batteries are widely used as source of direct current electrical energy in automobiles, boats, aircraft, portable electric and electronic equipment, and lightning. When a wire connects the two plates, electrical current flows between them. The simplest cells are made up of plates of two different kinds of metal, which are kept in salty or acid liquid. The battery is a combination of two or more electric cells. Duracell (Original Equipment Manufacturers pages).īatteries are simple devices. "Capacity may vary from 35 mAh for some of the small hearing-aid batteries to more than 100 Ah for a size 28 deep-cycle."Įnergy Density. Touch the probes to the battery under test.American Radio Relay League. Set the multimeter to the 200 milliamp scale. To use, just attach the alligator clip to one of the test leads. (Adding 19,000 ohms, if such a resistor were to be available, is more of a hypothetical example than a serious suggestion.) Here is an on-line calculator for parallel resistances. Resistances in parallel function differently than resistances in series. I could have brought the 367 ohms down to exactly 360 ohms by soldering another resistor of 19000 ohms in parallel to the two resistors. The cause of this lower voltage was chemical and it was considered a hard limit until some years ago. This greatly decreased their usability (many devices did not work well with 1.2 V). As the cell discharges products build up around the electrodes. I scraped the resistor lead opposite the alligator clip to remove any hot glue that might act as insulation. Until the last years, my knowledge was that the rechargeable analogue of the 1.5 V batteries have only 1.2 V. A new cell produces about 1.5 volts, but this differs considerably as the battery is used. I slipped a soda straw over them and added some hot glue at each end to provide some physical strength. I soldered a small alligator clip to one end of the resistors. The colors shown on the resistors in the illustration are accurate for the values indicated.) See the second photo. Also, resistors use a color coding system to indicate their value. My chain of resistors is actually 367 ohms. Xtar AA 1. Check the actual resistance with the Ohms function on your multimeter. (Resistors are usually not exactly their stated value, but vary just a little. Soldering these two end-to-end (series) gave me one resistance of about 370 ohms. 
I rummaged through my junk box and found a 270 ohm resistor and a 100 ohm resistor. It would be ideal if I could have gone to Radio Shack to buy a pack of 360 ohm resistors, but they are not available.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
